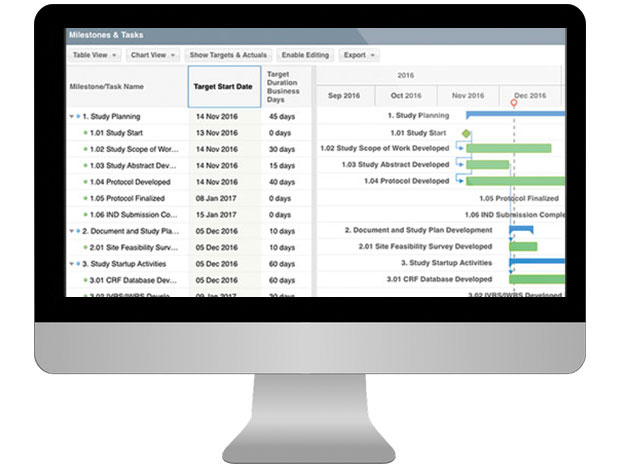
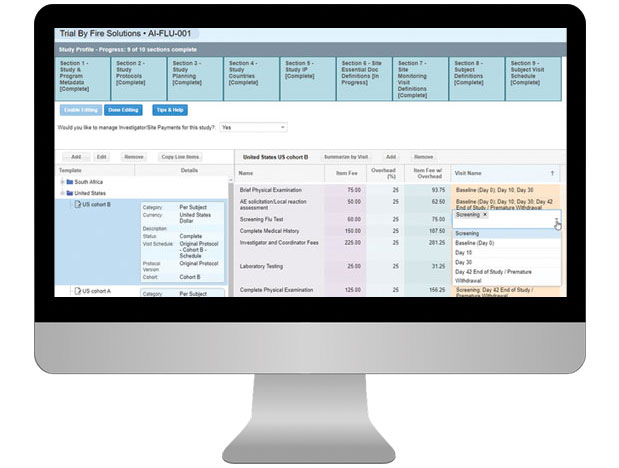
Electronic Visit Reports (EVRs)
Design EVR templates, specify appropriate EVR approvers, and author and approve EVR reports directly in the CTMS. View outstanding reports and oversee completion metrics for compliance with the monitoring plan. Utilize reminder and workflow alerts to guide on-time approvals. Specifically:
- Quickly create EVR templates per Sponsor requirements or internal templates
- Annotate CRA completion instructions directly in the template
- Easily setup Q & A sections, specify appropriate response, and indicate when a comment is required
- Build EVR templates using tables such as action items, deviations, IP review, ICF and data review, SAEs, and screening and enrollment
- Include confirmation and follow-up letters as EVR attachments to facilitate review and approvals
- Associate final EVR templates to studies for immediate use
- Use in-app and email alerts for EVR drafting through final approval; use alerts to manage due and nearly due reports for compliance with monitoring metrics
- Final approved e-signed reports and associated attachments are automatically posted in the eTMF