Please submit the form to access our product sheets:
Free Resource
Is Your TMF Inspection-Ready… or Inspection-Exposed?
Identify TMF gaps before inspectors do—and move from reactive audits to continuous inspection readiness.
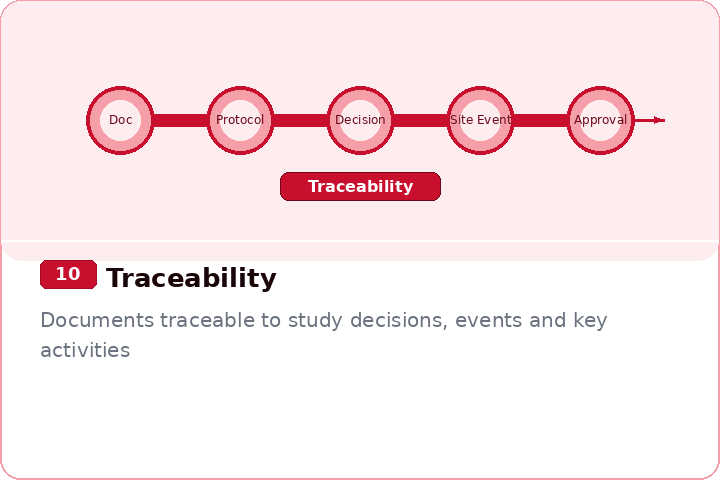
Assess completeness, traceability, and document quality
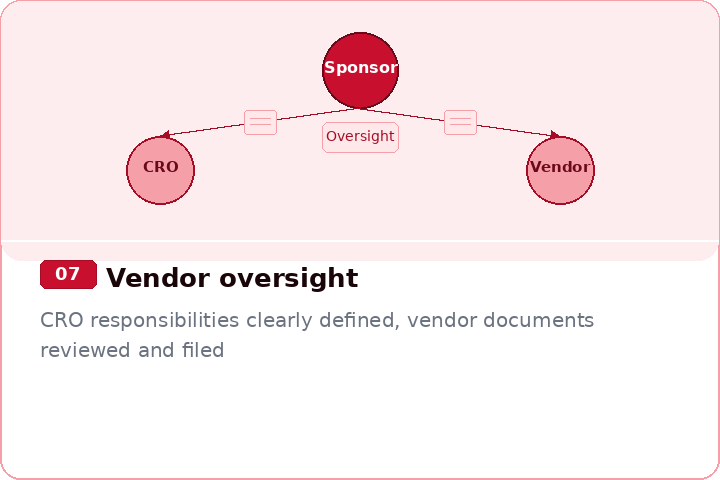
Prepare for FDA, Sponsor, and CRO Inspections
Strengthen QC, Ownership, and filing discipline
Covers all 14 inspection-readiness phases
Experience effective, user-focused study management solutions at an affordable price.
The Problem
Most TMFs don't fail until they're inspected
When an inspection is announced, teams discover gaps that could have been avoided. These issues create real compliance risk.
Our perspective
Inspection readiness isn't a milestone—it's a discipline
A Trial Master File isn't just a repository. It's the documented story of your clinical trial. Inspectors aren't simply asking whether documents exist.
"Can I reconstruct what happened in this trial—and trust it?"
That requires contemporaneous filing, clear document ownership, consistent QC across sponsors, CROs, and vendors, and complete audit trails that demonstrate accountability—not just activity.
What’s Included
14 phases of inspection readiness
Organized to follow the full inspection lifecycle—from first notification to post-inspection follow-up.
SimpleTrials eTMF
How SimpleTrials supports inspection readiness
Structured eTMF aligned with the TMF Reference Model
Role-based access control for secure collaboration
Complete audit trail and version history on every document
Standardized filing workflows for sponsors and CROs
Inspector-specific read-only access, pre-configurable
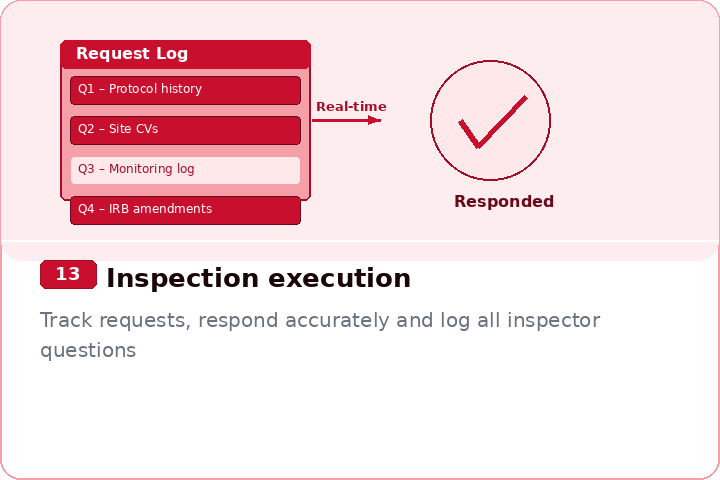
Faster document retrieval during live inspections
FAQ
Common questions
What is a TMF inspection checklist?
A structured tool used to assess whether a Trial Master File is complete, organized, and compliant with regulatory expectations before an audit or inspection.
What documents are required in a TMF?
Essential documents include regulatory approvals, site documentation, monitoring reports, safety reports, and trial management records as defined by ICH E6(R2).
What are the most common TMF inspection findings?
Missing or incomplete documents, poor document quality, lack of traceability, delayed filing, and inconsistent structure across sites and vendors.
How often should TMF reviews be performed?
Continuously throughout the study, with periodic QC and QA checks to maintain inspection readiness at all times—not just before audits.